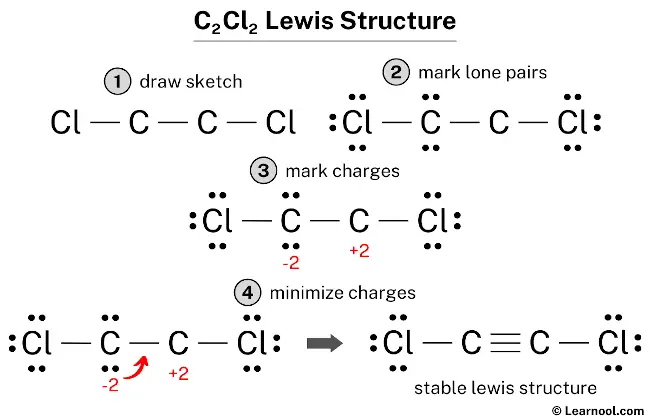
Scheme 3. Reagents and conditions: (a) ref. 21. (b) CH 2 Cl 2 , Lewis... | Download Scientific Diagram
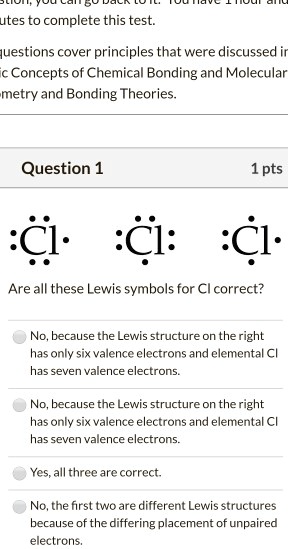
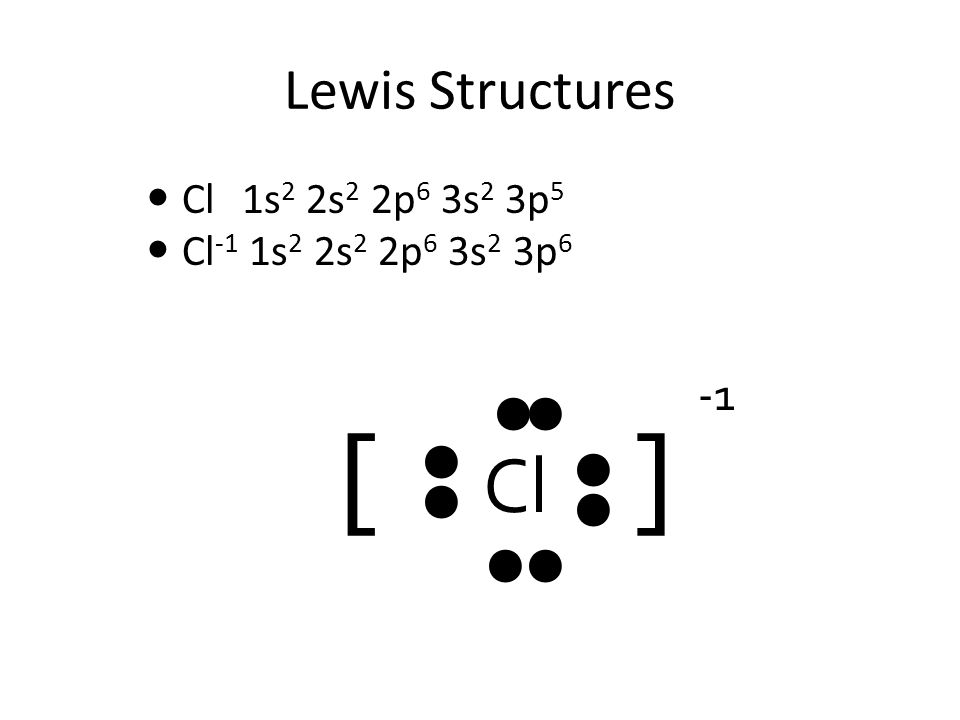
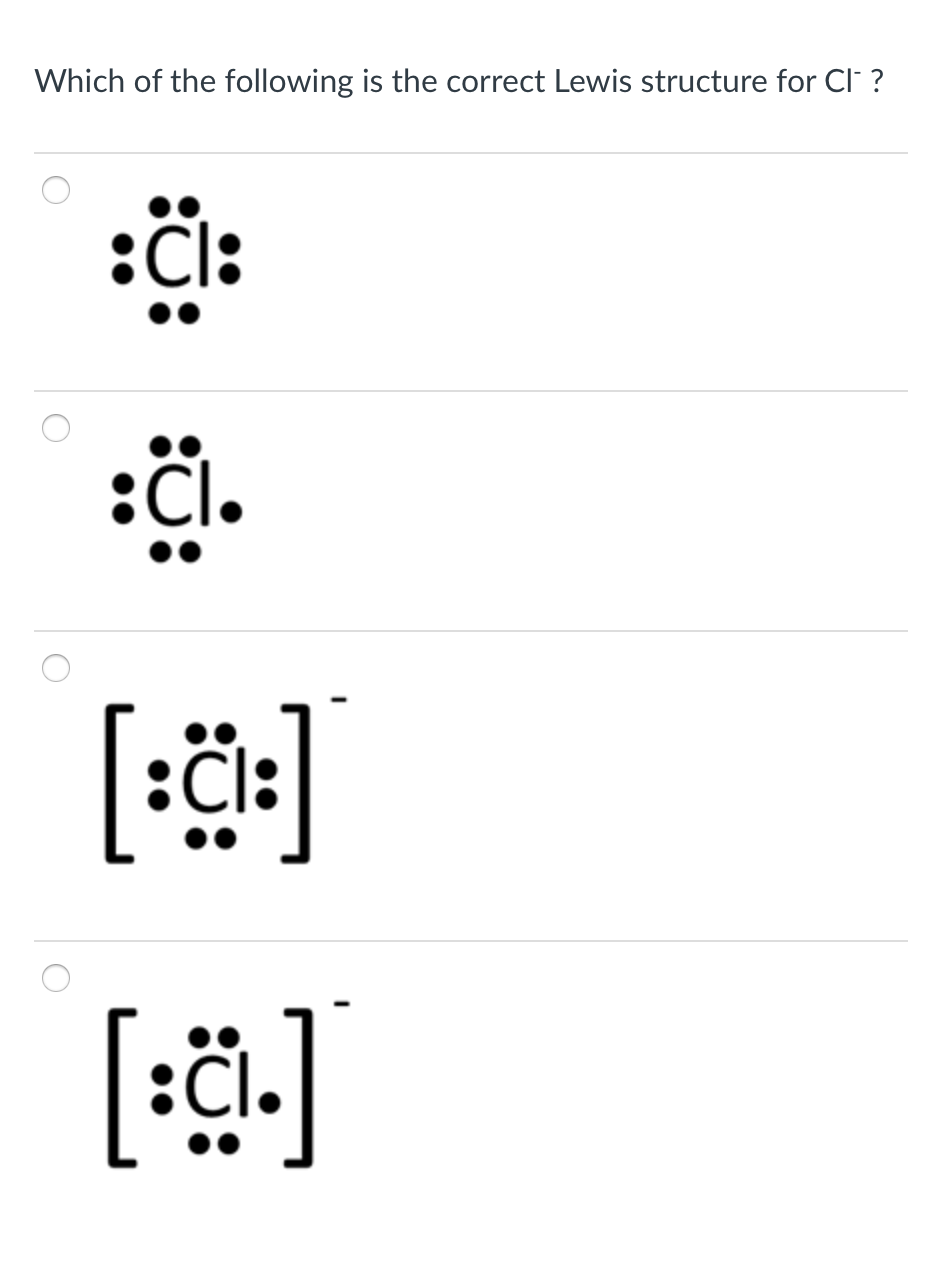
SOLVED:utes to complete this test: juestions cover principles that were discussed ir Concepts of Chemical Bonding and Molecular metry and Bonding Theories: Question 1 1pts Cl :Cl: 'Cl: Are all these Lewis

A handbook of the literature of the Rev. C.L. Dodgson (Lewis Carroll): Sidney Herbert Williams, Falconer Madan: Amazon.com: Books

SOLVED:Consider the two Lewis structures below: Based on formal charges, which structure is more likely (and why)? N=& N=0 The LEFT structure because oxygen is less electronegative than nitrogen_ The RIGHT structure